Following such data readout, we anticipate submitting a New Drug Application to the FDA to seek approval to commercially launch rigosertib.” Second Quarter .... Dec 21, 2020 — Notably, of the three currently approved CDK4/6 inhibitors, two are approved for ... Onconova's product candidate oral rigosertib is currently in a .... Jun 1, 2021 — Officials with the FDA have approved sotorasib (Lumakras; Amgen) as the first treatment for adult patients with non-small cell lung cancer ...
Sep 21, 2020 — A Phase 3 trial says rigosertib failed to prolong life in those with ... an approved medication of choice plus supportive care at prolonging the life ... Previous: FDA Grants Rare Pediatric Disease Status to AML Therapy OXi-4503.. Generic Name: rigosertib. Date Designated: 03/16/2012. Orphan Designation: Treatment of ovarian cancer. Orphan Designation Status: Designated/Withdrawn.. Rigosertib is not FDA-approved or commercially available. It is currently being used for research purposes only. The study doctor can explain how the study drug ...
rigosertib approval
rigosertib approval, rigosertib fda approval, rigosertib fda approval 2020, rigosertib fda approval 2021 Download Album edane the best of metal Mp3 (40:42 Min) - Free Full Download All Music
FDA-approved compounds and combinations into each well, in replicates, and incubates for a ... cell [blast] population) in primary MDS samples to rigosertib.. Jul 27, 2020 — Preclinical experiments with rigosertib in cellular models ... and health authority approved agents are ongoing worldwide to seek to address the .... rigosertib fda approval 2020. By. In Sem categoria. Posted jun 4, 2021. […] 2016 Apr;17(4):496-508. doi: 10.1016/S1470-2045(16)00009-7. NEWTOWN, Pa.. Jun 17, 2021 — Rigosertib synergistically combined with immune checkpoint blockade ... On Tuesday, Sorrento announced that the FDA has given the green light for ... adults to test the candidate and hope to get approvals by the end of 2021. The Caligula Effect: Overdose - Digital Art Book Download Game Hacked

rigosertib fda approval 2020
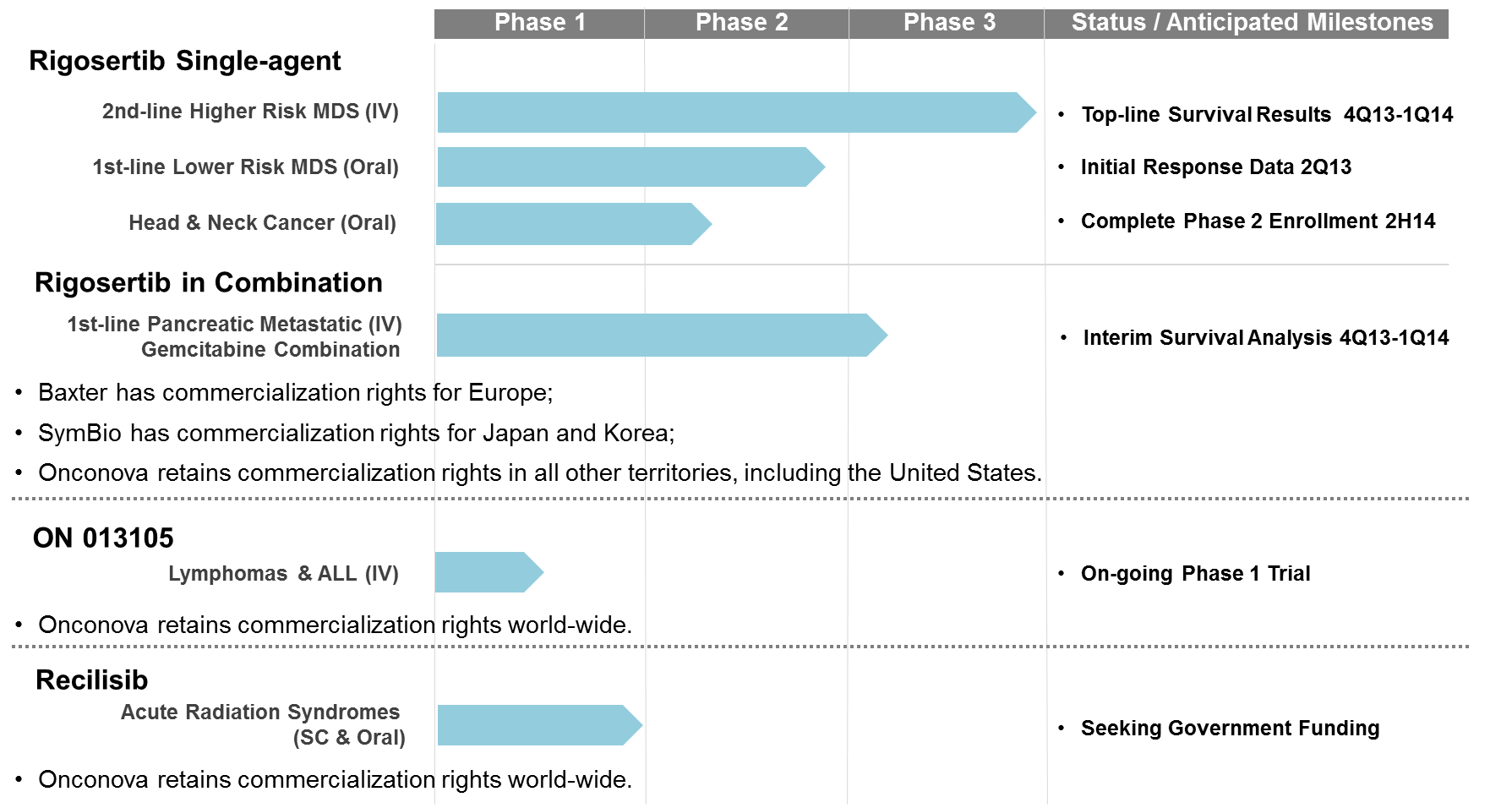
Onconova Therapeutics Announces FDA Permission For Study To Proceed Under Its ... ONCONOVA THERAPEUTICS INC - IV RIGOSERTIB DID NOT MEET ... Onconova Therapeutics Says Compensation Committee Approved A Cash Bonus .... Rigosertib has been granted orphan drug status for MDS in both the United States ... under the FDA's Animal Efficacy Rule, which permits marketing approval for .... The Chinese IND was approved in January 2020. ... myelodysplastic syndromes (“HR-MDS”) and 50% upon U.S. FDA approval of rigosertib for HR-MDS. Montana Behind The Scenes Ebook Rar

FDA has granted Orphan Drug Designation for the use of rigosertib in MDS. The clinical program in solid tumors is also advancing with initiation of the Phase II/III .... Feb 27, 2020 — In addition the company is also involved in using rigosertib together ... burning millions of dollars with no MDS drug approved by the FDA yet.. Onconova Announces Successful End-of-Phase 2 Meeting with FDA for Oral ... Drug Administration (FDA) for the combination of oral rigosertib with azacitidine for ... eligible 1st-line patients, were approved by the FDA more than a decade ago.. Jan 2, 2019 — ... (SPA) to FDA for Phase 3 Trial of Oral Rigosertib in Combination with ... requirements for a study to ultimately support marketing approval.. In December 2020, we received clearance from the FDA to begin the study, and have since received IRB approval at our first site. Onconova Therapeutics shares .... Potential to overcome resistance to approved CDK 4/6 inhibitors in HR+ HER 2- ... Rigosertib is in multiple investigator-sponsored combination studies. ▫. a0c380760d I should have asked Santa for boys instead of a puppy, 78463AD9-6A74-4E07-9710-8DF77686 @iMGSRC.RU
